Development of a summary framework for communication of payer needs
A conceptual summary framework of payer requirements to guide development of pharmaceuticals from the payer perspective
The challenge
The silo mentality within pharmaceutical companies was recognised as a barrier to building in value from the payer perspective during the early stages of drug development/acquisition. This could lead to products reaching late stage development without value from the payer perspective ‘built in’.
The solution
In order to successfully commercialise and launch innovative healthcare products, an evaluation and communication framework must be in place to assess payer requirements at each step of the development lifecycle. The following activities were conducted to achieve this:
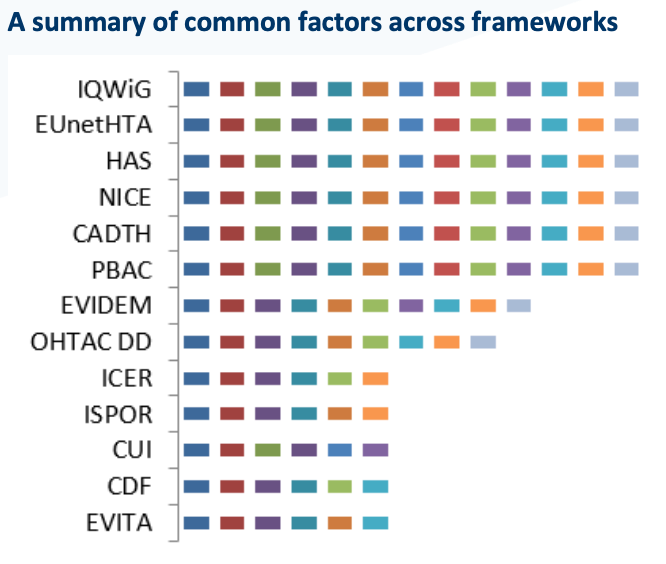
> A structured literature review, including a review of HTA requirements and grey literature sources, to identify existing frameworks and perspectives on frameworks
> Evaluation of existing HTA and non-HTA frameworks to identify common criteria of importance to the payer
> Development of a summary framework based on the identified key elements
> Development of a mnemonic to ensure the key factors were easily remembered and used in practice
> Defined case studies to test the application of the framework
> Development of a communications plan to introduce the framework to the pharmaceutical industry.
The development of the framework was a collaborative effort between Adelphi Values PROVE, our client and a steering group of leading health economists and HTA experts.
Key results
The summary framework was published in a peerreviewed journal and presented to the pharmaceutical industry and healthcare stakeholders via Market Access and Health Economics and Outcomes Research conferences.
BEACON: A Summary Framework to Overcome Potential Reimbursement Hurdles. PharmacoEconomics 34, 1051– 1065 (2016).
https://doi.org/10.1007/s40273-016-0427-7
Value to the client
The framework was implemented and utilised by our client’s cross-functional team to improve communication and to drive value-based decision making for the assessment, acquisition or development of pharmaceutical products. The framework was used to position our client as leaders in Market Access externally to payers, academics, potential partners and candidates.